Module 4 Problem Set Quiz
Module 4 Problem Set
-
Write electron (subshell) configurations for the following atoms:
19K
35Br
Correct Answer

Write orbital diagrams for the following atoms:
15P 28Ni
Correct Answer
ORBITAL DIAGRAMS
15P = 1s2 2s2 2p6 3s2 3p3
↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑ ↑ ↑
1s 2s 2p 3s 3p
28Ni = 1s2 2s2 2p6 3s2 3p6 4s2 3d8
↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑↓ ↑ ↑
1s 2s 2p 3s 3p 4s 3d
Write the values for the four quantum numbers for a 4d8 electron.
Correct Answer
Orbital diagram for the 4d8 orbital
↑↓↑↓↑↓↑↑
A 4d8 electron is in the 4th shell so n = 4. Its "d" orbital has 2 nodal planes so l = 2. The electron is in the 3rd of the orbitals of the d subshell so ml = 0. And since this is the 2nd electron to occupy this "d" orbital, ms = -1/2. In summary, the values for the four quantum numbers for the 4d8 electron are: n = 4, l = 2, ml = 0 and ms = -1/2.
Write the values for the four quantum numbers for the last electron to fill 28Ni.
Correct Answer
Electron configuration for Ni
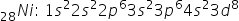
Orbital Diagram for 3d orbital is
↑↓↑↓↑↓↑↑
The last electron to fill 28Ni is a 3d8 electron which is in the 3rd shell so n = 3. Its "d" orbital has 2 nodal planes so l = 2. The electron is in the 3rd of the orbitals of the d subshell so ml = 0. And since this is the 2nd electron to occupy this "d" orbital, ms = -1/2. In summary, the values for the four quantum numbers for the 3d8 electron are: n = 3, l = 2, ml = 0 and ms = -1/2.
Choose the atom that matches the identified atomic property and explain your answer based on the periodic trend and its position within a group or period in the periodic table.
Highest Ionization Energy: Li, Be, B
Correct Answer
B has the highest ionization energy since it is farthest to right of these elements and ionization energy increases as you go from left to right in a period.
Choose the atom that matches the identified atomic property and explain your answer based on the periodic trend and its position within a group or period in the periodic table.
Lowest Electronegativity: Li, Be, B
Correct Answer
Li has the lowest electronegativity since it is farthest to left of these elements and electronegativity increases as you go from left to right in a period.
Choose the atom that matches the identified atomic property and explain your answer based on the periodic trend and its position within a group or period in the periodic table.
Largest Atomic Size: Li, Be, B
Correct Answer
Li has the largest atomic size since it is farthest to left of these elements and atomic size decreases as you go from left to right in a period.
Create an orbital diagram and Lewis dot structure for 16S.
Correct Answer
Electron configuration of S
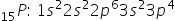
Orbital diagram for S
↑↓↑↓↑↓↑↓↑↓↑↓↑↓↑↑
1s 2s 2p 3s 3p
Lewis structure
From the orbital diagram, there are 2 unpaired electrons in the 3p orbital. Thus, the Lewis structure of S is:

